VR developed by Albert Liu, Kate Jensen, Chih-Mei Young and Theodore Hall
Extra controls/functions:
- Move the particles: Place your pointer (the small tetrahedra) on the surface of the particles until it shows a four-sided star-like symbol (see the right figure below, on the surface of the right particle). Use the front trigger of your controller to grab the particle and move it.
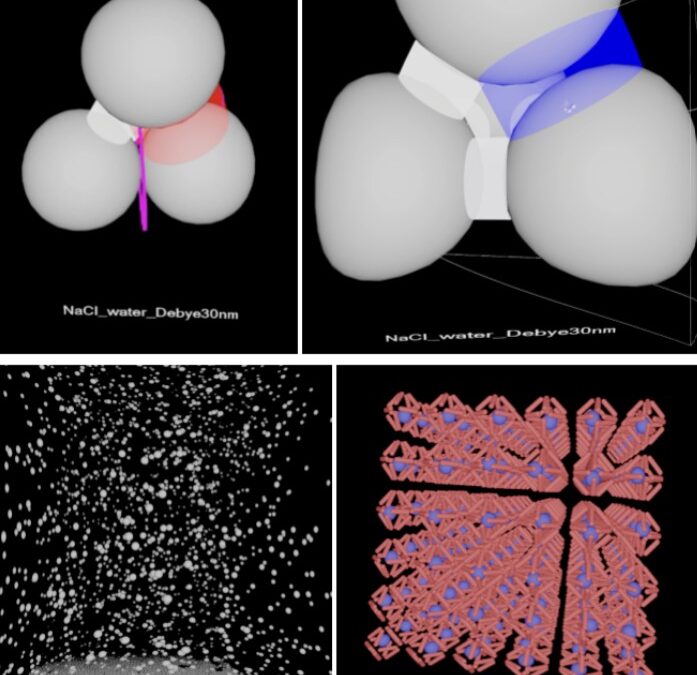
- Change the Debye lengths (3 options): You can use <Y> button on the left-hand controller, or the function key <F6> on the keyboard (make sure the VR window in the computer is clicked), to cycle forward or backward through the Debye lengths (these relate to cycling through different solvents). As you do, you may see how the cylinders change in diameter and color especially when the interparticle distance is small.
- Cylinder diameter: The cylinder diameter is scaled with the potential energy. If the particles’ inter-surface distance gets too close to zero or negative (intersecting), it appears as a magenta ring or “halo” (see the left figure below, for the bottom particles).
- Cylinder colors: The color saturation is scaled with the force between particles, the color hue corresponds to the attraction/repulsion (red corresponds to attraction, blue corresponds to repulsion, white corresponds to zero-force). This means that darker blue corresponds to higher repulsive force, and darker red corresponds to higher attraction force than lighter (closer to white) colors.
- Switching between surface and centroids views: You can switch between two different views using the <X> button on the left-hand controller or the function key <F7> on keyboard (make sure the VR window in the computer is clicked when using the keyboard).
The third module aims to help students understand the interactions between particles and their surroundings. It consists of three activities: (1) Interparticle Interactions, (2) Electric Field Assembly, and (3) Cage Diffusion.
Activity 1: Interparticle Interactions
This activity introduces particle interactions governed by DLVO theory, including electrostatic and van der Waals forces. The system consists of particles with a zeta potential of −30 mV in an aqueous solution containing a monovalent salt. Students can manipulate the interparticle distance and observe how interaction forces change as a function of separation distance and Debye length. Pairwise interactions are visualized using connecting cylinders (not physical bonds). This activity helps students identify equilibrium structures and understand how colloidal stability depends on competing forces
Activity 2: Electric-field assembly of particles
This activity explores the electric-field-driven self-assembly of colloidal particles. When a DC electric field is applied, charged particles undergo electrophoresis toward an oppositely charged electrode and subsequently assemble into ordered crystal structures. Students can compare the effects of high and low electric field strengths on the resulting structures, including differences in defect formation and ordering. These structural variations influence material properties such as optical behavior (e.g., structural color)
Activity 3: Cage diffusion
This activity examines particle dynamics under confinement using a cage-like environment. Nanoparticles undergo Brownian motion, but their movement is restricted within octahedral cages simulated via molecular dynamics. Students can visualize 3D trajectories for particles of different sizes and compare confined versus escaped behavior. The activity emphasizes how particle size influences diffusion dynamics and escape time from the cage structure
We also have a CAVE version of Interparticle Interactions for the students to compare the experience across different visualization technologies.